Laws of Thermodynamics
The four laws of thermodynamics are the fundamental principles which define the relationship between Temperature, Energy and Entropy. The four laws describe how these interact in different scenarios and systems. The laws also forbid impossible scenarios such as perpetual motion. The four laws of thermodynamics are:
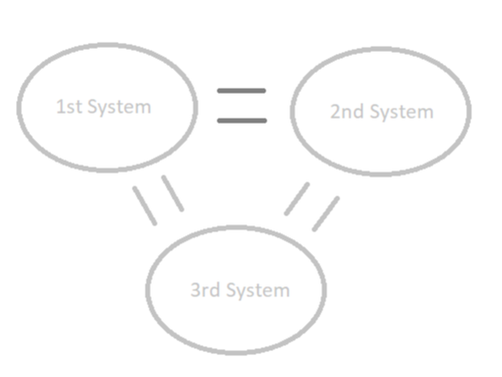
Zeroth law of ThermodynamicsZeroth (0th) Law of Thermodynamics: the zeroth law of thermodynamics states that if ta first and second thermal system are in equilibrium with a third system, then by default they are in equilibrium with each other. The Image to the right shows three systems (circled). the first and third are in equilibruim, as are the 2nd and third system. As a result, the first and second system must be in equilibrium also. |
First law of Thermodynamics
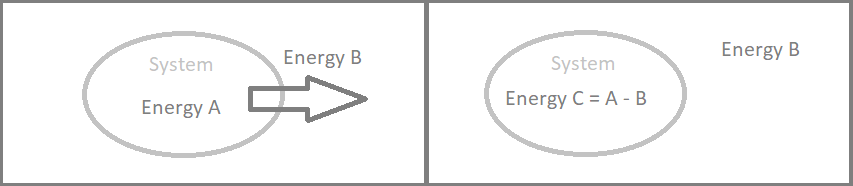
First (1st) Law of Thermodynamics: This Law is the conservation of energy law. In simplest terms, when energy leaves a system, whether it be work, heat or matter, the internal energy of the system must reduce by the same amount.
The Image above displays this laws as two stages of a system. The stage on the left shows a system at Energy A, with Energy B leaving the system. The second stage shows a system with Energy C, which is equal to Energy A, Less Energy B. Thus the net energy continues to be Energy A, and the conservation of energy is retained.
Second law of ThermodynamicsSecond (2nd) Law of Thermodynamics:the second law of thermodynamics states that the sum of the entropy of the systems increases during a natural thermodynamic process.
Third law of Thermodynamics |
|
Third (3rd) Law of Thermodynamics: the third law states that the Entropy of a system must be at it's minimum value at absolute zero. Fundamentally meaning that a for a perfect crystalline structure at absolute zero, the energy of the system is also zero. If a system is not a perfect crystal, then as a system approaches absolute zero (0 Kelvin), the system must be in the state with the minimum energy.